Start with the workflows that matter most
Coordinators currently track ConMed–AE relationships manually, across disconnected systems. Neem automatically surfaces potential relationships, flags them for review, and prepares documentation — with the coordinator confirming every step.
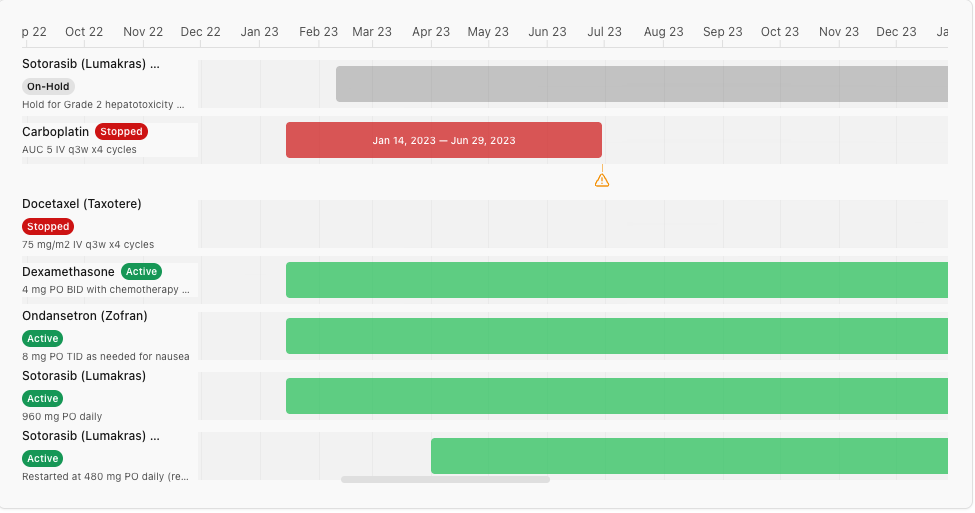
Missed ConMed–AE relationships are among the most common sources of protocol deviations and safety reporting failures. This is one of the highest-stakes workflows in trial operations, and one of the most manual. Neem changes that without removing the human from the loop.